The Road to the Pill: Designing, Making, and Sustaining Solid Orals
Solid oral dosage forms are the most familiar way of taking medicines. A tablet or capsule is easy to carry, simple to take, and usually cheaper to make at scale. From a development point of view, they also give good control over dose accuracy, quality, and shelf-life. For many drugs, a well-designed solid oral product can balance patient convenience with robust manufacturing and regulatory compliance.

Why do companies prefer solids? First, stability. Most actives are more stable in the dry state than in solution, so we can achieve longer shelf-life with simpler packaging. Second, manufacturability. Modern tableting and encapsulation allow high throughput with tight in-process controls. Third, flexibility. Using coatings, polymers, and multiparticulate designs, we can tune when and where the drug releases in the GI tract—immediate, delayed, extended, or even targeted to the colon. Finally, access. Solids are cost-effective to ship and store, important for public health programs and private markets alike.
However, solid orals are not “one size fits all.” We still need the right match between drug properties, patient needs, and business goals. Poorly soluble drugs may need special approaches (salts, amorphous dispersions, particle engineering, or lipids). Acid-labile or gastric-irritant drugs may need enteric protection. Highly potent molecules need containment and dose uniformity at very low strengths. Pediatric and geriatric users may struggle with swallowing; here, orodispersible tablets, sprinkles, or mini-tablets can help. For chronic diseases, extended-release can improve adherence by reducing dosing frequency. For acute indications, rapid disintegration and fast onset may be more valuable than once-daily convenience.
From a quality standpoint, solid orals are governed by well-defined compendial tests and ICH expectations: identity, assay, impurities, content uniformity, dissolution, disintegration, hardness/friability (as relevant), microbial limits (when needed), stability under ICH zones, and extractables/leachables for packaging. Early formulation work typically combines preformulation (solubility, pKa, hygroscopicity, polymorph form), excipient screening, and manufacturability checks (flow, compressibility). Process selection—direct compression, dry granulation, or wet granulation—follows the powder’s behavior and the product’s release design. Risk assessment (e.g., QbD) helps define critical material attributes and process parameters; scale-up uses design of experiments, robust controls, and meaningful dissolution methods.
In short, solid oral dosage forms remain the backbone of modern therapeutics because they can be engineered to meet many clinical, technical, and commercial constraints. The “art” lies in choosing the right platform—tablet, capsule, multiparticulate, or a hybrid—and then building a simple, stable, and scalable product around the active.
Connect with our scientific experts for your drug discovery, development and manufacturing needs
Dosage Forms
What they are
A dosage form is the “finished shape” in which a medicine is delivered to the patient. It is how an active pharmaceutical ingredient (API) is combined with selected excipients and processing steps to become a usable product with a defined dose, quality, and shelf-life. Tablets, capsules, injections, eye drops, inhalers, ointments, and patches are all dosage forms. Each form is designed to protect the drug, present the right amount to the body, and make administration safe and acceptable for the intended user. In development, the dosage form links science to real-world use: it translates the molecule’s properties into a product that can be manufactured reliably, stored and shipped without loss of quality, and taken correctly by patients.
Why do we need different dosage forms?
Different dosage forms exist because drugs and patients are diverse, and clinical needs vary. Some molecules are unstable in water and survive better in dry, solid formats. Others need to act very fast and therefore are given as injections or rapidly dissolving oral products. Certain drugs irritate the stomach and need enteric protection to release only in the intestine. Children and older adults may struggle to swallow, so a dispersible tablet, mini-tablet, or an orodispersible film can improve acceptance. For chronic diseases, once-daily modified-release tablets help adherence; for acute conditions, immediate-release dosing with quick onset may be more appropriate. Beyond patient and pharmacology factors, manufacturing, packaging, storage conditions, and cost of goods also influence the choice. A good dosage form balances the API’s physicochemical profile, the pharmacokinetic target, patient convenience, regulatory expectations, and commercial viability.
How are dosage forms classified?
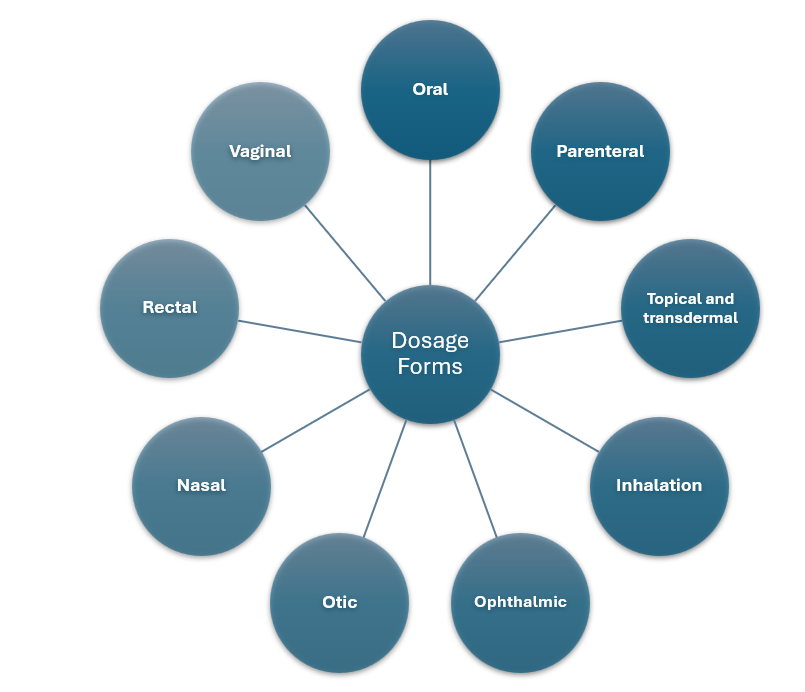
There are many ways to classify dosage forms. A practical view starts with the route of administration.
Oral products include solids like tablets and capsules, as well as liquids like solutions and suspensions.
Parenteral products are injections and infusions delivered intravenously, intramuscularly, or subcutaneously when rapid action, precise bioavailability, or bypassing the gut is needed.
Topical and transdermal products act on or through the skin for local relief or systemic delivery over time.
Inhalation products deliver to the lungs for fast systemic action or local respiratory therapy.
Ophthalmic, otic, and nasal forms treat eyes, ears, and nasal passages with strict purity and compatibility requirements.
Rectal and vaginal dosage forms can offer local therapy or an alternative route when oral dosing is not feasible.
Within each route, forms are further grouped by release behavior—immediate-release, delayed-release, and extended-release—because where and how fast the drug is released strongly affects efficacy and safety.
Solid oral dosage forms
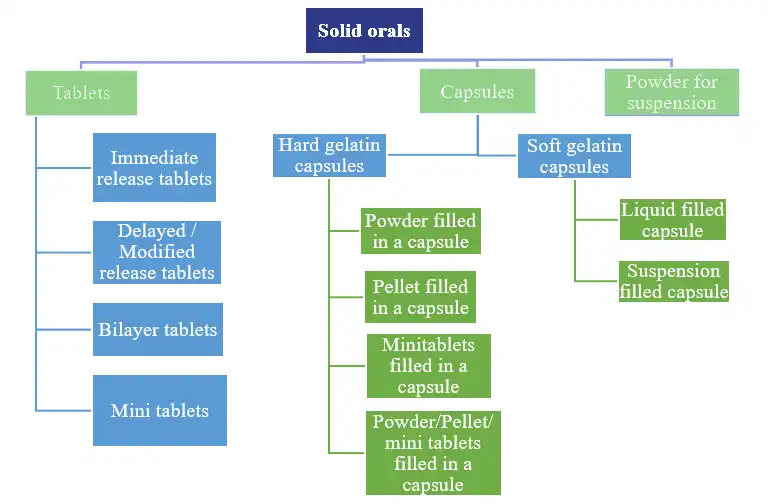
Solid oral dosage forms remain the backbone of therapy because they are simple to use, stable in the dry state, and efficient to manufacture at scale. The most common are tablets and capsules, but the family is broad and flexible, allowing precise tailoring of performance to clinical goals.
Immediate-release tablets are the standard swallow tablets designed to disintegrate and dissolve quickly in the gastrointestinal tract. They suit a wide range of small-molecule drugs where rapid onset and straightforward exposure are desired. Developers pay attention to powder flow, compressibility, and dissolution in physiologically relevant media so that in-vivo exposure is consistent. Film coating is often added to improve swallowability, protect against moisture and light, and mask taste without changing the core release profile.
Chewable and dispersible tablets adapt the same principles for populations who need easier administration. Chewables are formulated for pleasant mouthfeel and robust taste-masking so that the tablet can be chewed before swallowing. Dispersible or soluble tablets are intended to be mixed with water to form a suspension or solution; these are helpful for dose flexibility in pediatrics and for patients with dysphagia. Control of dispersion time, sediment behavior, and palatability is central to their success.
Effervescent tablets and granules contain acid–base pairs that release carbon dioxide when added to water. The bubbling action aids rapid dispersion and can carry large doses in a palatable drink. Because they are highly moisture-sensitive, packaging uses tight barriers like aluminum tubes with desiccants. These formats are popular for electrolytes, vitamins, and some analgesics where fast, drinkable delivery is valued.
Orodispersible tablets (ODTs) and orodispersible films (ODFs) are designed to disintegrate on the tongue without water, usually within half a minute. They are useful for patients who travel, who experience nausea, or who need discreet dosing. Formulation leans on porous matrices and super-disintegrants, combined with strong taste-masking because the API is exposed to taste buds before swallowing. Moisture-barrier blister packs protect these fragile forms during storage and handling.
Modified-release tablets change when and where the drug is released. Extended-release products slow the release over 8–24 hours using hydrophilic matrices, lipid matrices, or multiparticulate systems compressed into tablets or filled into capsules. The goal is smoother exposure with fewer peaks and troughs, enabling once-daily dosing and better adherence. Delayed-release (enteric) products resist stomach acid and release downstream in the intestine, protecting acid-labile APIs or avoiding gastric irritation. Development focuses on selecting polymers with the right pH triggers, validating multi-stage dissolution methods, and building in-vitro/in-vivo links that predict performance.
Hard-shell capsules made of gelatin or HPMC offer a versatile platform. They can be filled with blended powders, pellets, mini-tablets, or even certain liquids with appropriate sealing. Capsules are particularly useful for low-dose potent drugs because they support good content uniformity and quick scale-up in early development. HPMC shells help with moisture-sensitive actives and allow vegetarian positioning. Because the shell dissolves promptly, the release profile is controlled mainly by the fill—whether it is an immediate-release blend or a coated multiparticulate.
Soft gelatin capsules (softgels) enclose a liquid or semi-solid fill in a flexible shell. They are effective for poorly soluble, lipophilic drugs that benefit from self-emulsifying or solubilized fills to enhance bioavailability. Softgels also improve patient acceptance for bulky or oily actives. Development must address shell–fill compatibility and ensure the fill remains stable and homogeneous over shelf-life.
Powders, granules, pellets, and mini-tablets extend the solid oral toolkit. Unit-dose sachets or multi-dose jars of powders and granules provide flexible dosing, especially for pediatrics or high-dose electrolytes and antibiotics. Pellets (small spheres) and mini-tablets (1–3 mm) can carry different coatings to create complex release profiles. They are usually filled into capsules or stick-packs and often empty from the stomach more uniformly than a single monolithic tablet, reducing variability. For combination therapy, mixing multiple coated populations in a single dose allows staged or differential release without chemical incompatibility.
Across all these solid oral formats, a few themes are constant. Patient-centric design drives choices in size, swallowability, taste, and dosing frequency. Quality is demonstrated through well-established tests for identity, assay, impurities, content uniformity, and dissolution that reflect how the product will perform in the body. Stability studies under the intended climate zones and suitable packaging—HDPE bottles with desiccants, alu-alu blisters, tubes, or pouches—protect the product through its life. Finally, the manufacturing route—direct compression, dry granulation, wet granulation, extrusion–spheronization, or encapsulation—is chosen to fit the powder properties and the desired release behavior while remaining efficient and scalable.
In summary, dosage forms are the practical face of pharmaceutical science, and solid orals are the most versatile expression of that practice. By understanding the reasons behind the form—molecule needs, patient needs, and system constraints—we can choose and engineer a solid oral product that is simple, stable, and truly usable in everyday healthcare.
What Aurigene offers
Aurigene’s oral solid dosage capabilities are presented as a complete, end-to-end offering: advanced, US-FDA–inspected facilities and equipment are deployed to develop and scale tablets, capsules, powders, and multiparticulates; structured services cover pre-formulation through CMC, stability, scale-up, BE enablement, and tech transfer to commercial; and proven specialties—modified/enteric release, multiparticulate design, taste-masking, and patient-centric formats—are delivered by a seasoned team with decades of formulation experience.
- US-FDA–inspected lab and manufacturing facilities.
- State-of-the-art oral solid suites with low-humidity areas and controls for light-sensitive compounds.
- Equipment platforms: direct tablet compression; fluid-bed granulation/drying/coating; high-shear wet granulation; dry granulation (roller compaction, slugging); extrusion–spheronization; high-speed automated tablet presses; perforated pan coaters (including mini-tablet coating); automated capsule filling for powders, granules, pellets, and mini-tablets.
- Scale range: ~500 to 50,000 units at R&D/pilot scale; up to ~7,500,000 units at commercial sites.
- Fit-for-Purpose formulation development for early phase and First-in-Human studies, extended through to commercial manufacturing.
- Prototype screening, formulation and process optimisation using QbD principles, and ICH-compliant stability studies.
- Scale-up, technology transfer, and complete CMC authoring/support for regulatory submissions.
- Development of oral solid dosage forms across tablets, capsules, powders for suspension, and multiparticulate systems tailored for immediate-, modified-, and targeted-release.
- Tablet formats: immediate-release, delayed-/enteric-release, sustained/extended-release, orally disintegrating, film-coated, taste-masked, sublingual, buccal, effervescent, chewable, fixed-dose combinations, bilayer/trilayer, and mini-tablets (including MUPS).
- Capsule formats: hard-gelatin (or equivalent) filled with powders, pellets, and/or mini-tablets in combinations; soft-gelatin capsules with liquid/suspension fills for poorly soluble or thermo/photo-sensitive actives.
- Pediatric-friendly powders for oral suspension (age-appropriate, easily reconstitutable), including sachet presentation.
- Custom formulation support for veterinary and nutraceutical applications.
- 20+ years of formulation experience with advanced oral technologies.
- Proven expertise with acid-labile APIs, enteric protection, GI-region targeting, and modified-release design.
- Strong capability in multiparticulate systems (pellets/mini-tablets/MUPS) to reduce dose dumping and tailor release.
- Taste-masking and patient-centric design for swallowability, pediatric/geriatric acceptance, and user experience.
- Lifecycle coverage proclaimed “across the product lifecycle,” from FIH enablement through commercial supply, with audit-ready documentation and transfer packages.
- Highly skilled scientific team experienced across simple and complex oral solids.
Challenges for solid oral dosage forms
Poor solubility and permeability
Many new drug candidates fall into low-solubility classes. Simple tablets are then not enough; particle engineering, amorphous dispersions, or lipid systems are often needed. Each option brings extra steps, stability questions, and cost. Reliable bioavailability must still be achieved across fed/fasted states and variable GI physiology.
Stability in hot and humid climates
In many markets, high temperature and humidity are routine. Hygroscopic APIs and excipients take up moisture, leading to soft tablets, dissolution drift, or microbial risk for certain formats. Robust packaging and desiccants help, but pack size, cost, and patient convenience get affected.

Content uniformity at low dose and HPAPI handling
Dose strengths are moving lower as potency increases. Micro-dosing requires extremely uniform blends and tight process control. Containment for highly potent APIs raises capital needs, training demands, and cleaning validation complexity.
Manufacturing robustness
Practical defects—sticking, picking, capping, and lamination—still interrupt production. Powder flow varies lot to lot; excipient variability adds another layer. Scale-up from pilot to commercial remains non-trivial, especially for multiparticulates and coated systems that are sensitive to subtle changes in shear and humidity.
Reliable dissolution methods and IVIVC
For modified-release and enteric products, simple single-medium dissolution is rarely predictive. Multi-stage, biorelevant methods are needed, but these must remain discriminating and manageable for routine QC. Establishing IVIVC/IVIVR is valuable yet not always feasible.
Regulatory expectations and data integrity
Expectations continue to rise: nitrosamine risk assessments, extractables/leachables, lifecycle change management, and electronic records that hold up to audit. New guidance appears; global alignment is improving but not perfect, so multi-region programs carry extra documentation work.
Patient-centricity and adherence
Large tablets, bitter actives, and high daily pill burdens reduce adherence. Pediatric and geriatric populations need smaller units, better taste, and simpler instructions. Formulation solutions exist, but they add development effort and must still meet cost targets.
Supply chain and sustainability
Excipient shortages, variable quality, and long lead times disrupt plans. Sustainability targets push for solvent reduction, energy-efficient drying, lighter packs, and improved recyclability, all without compromising protection in humid zones.
Future outlook for oral solids
More enabling technologies, used earlier
Amorphous solid dispersions, hot-melt extrusion, spray drying, and nano-milling are likely to be evaluated during lead optimisation, not only as rescue tools later. This shift should shorten timelines by matching molecules to feasible oral platforms from day one.
Rise of multiparticulate and mini-tablet designs
Pellets and mini-tablets permit fine control of release, mixing of multiple profiles in one dose, and better gastric emptying. Their use is expected to grow in pediatrics, in fixed-dose combinations, and in programs that need flexible titration without reformulating the core.
Biorelevant dissolution and model-informed development
Wider use of physiologically based biopharmaceutics modelling (PBBM) and better in-vitro methods is anticipated. Dissolution tests will be designed with in-vivo relevance in mind, improving specification setting and supporting biowaivers where appropriate. Real-time data analysis should help detect drift early and avoid clinical surprises.
Continuous manufacturing and real-time release testing
Continuous direct compression and continuous coating are moving from pilots to production. With PAT tools (for example, NIR for blend uniformity), tighter control of variability can be achieved and real-time release can be enabled in defined cases. The benefits should include smaller footprints, faster changeovers, and more resilient supply.
Advanced coatings and targeted delivery
Smarter enteric and time-dependent coatings are expected to improve targeting to specific gut regions, including the colon. This will support drugs that are acid-labile, locally acting, or best absorbed distally. Better control of coating thickness distribution will reduce batch-to-batch variability.

Personalised and adaptive dosing
True one-patient-one-dose remains challenging at scale, but progress is being made. 3D printing for niche indications, modular mini-tablet kits for up-titration, and pharmacy-level compounding guidance for dispersible or sprinkle formats are likely to expand. Digital tools that connect dosing history with refill packs could make personalised schedules practical without changing the core formulation.
Taste-masking and sensory science
As more pediatric and geriatric programs move oral, structured sensory work will be used earlier. Coating, ion-exchange resins, microencapsulation, and sweetener systems will be combined with human factor studies to balance palatability and rapid disintegration.
Greener processes and packs
Solvent-free granulation, energy-efficient drying, and lower-temperature coating are expected to be adopted more widely. Packaging will move toward better barrier-to-weight ratios and improved recyclability, while still protecting against humidity. Life-cycle assessments will guide material choices.
Sharper quality systems and digital QMS
QbD will be the default, not a differentiator. Electronic batch records, deviation/CAPA analytics, and integrated stability LIMS will be standard. AI-assisted trending of IPC data should support predictive maintenance, early-warning for compression issues, and faster root-cause analysis.
Complex generics and hybrids
Growth is expected in complex oral products such as abuse-deterrent formulations, combination MR profiles, and enteric-protected micro-units. Hybrid pathways (similar to 505(b)(2)) will be used to bring incremental innovation—new strengths, new dose forms, and new release profiles—without the burden of full NCE programs.
What will not change
Solid orals will continue to be preferred where possible because they are stable, easy to handle, and cost-effective. The fundamentals will remain the same: the API must be understood in the solid state; a simple, robust process must be chosen; dissolution must reflect what happens in the body; and the product must be easy for patients to take and for plants to make. The future will add better tools and smarter data, but the core craft of making a reliable tablet or capsule will stay at the centre of therapy.




















