Experience and expertise are key to successful new drug development. There are various stages to a drug development program that need alignment of objectives and optimization of resource utilization. While the clinical success or failure of a drug is majorly influenced by its therapeutic potential, how systematically it is developed has an equal contribution to its success.
By integrating the development and manufacturing of drug substance and formulations, we are uniquely positioned to offer a significant advantage to customers that leverage the technical integration of both teams. 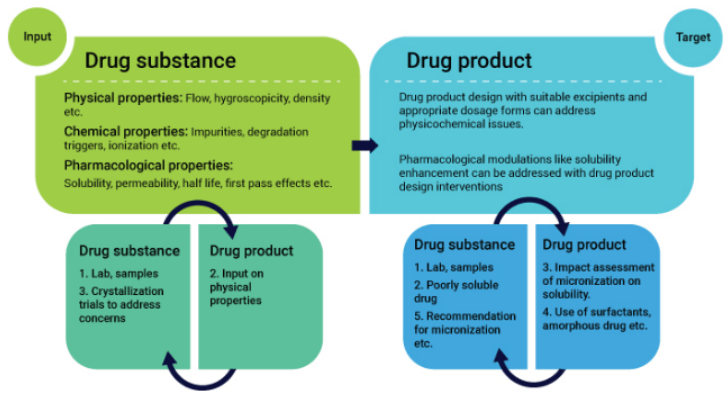



Why Aurigene Integrated Drug Substance and Drug Product (DS-DP) Services?
20+ years legacy of providing integrated API and formulation services
Fit-for-purpose development strategy
Co-located cross-functional teams
Services across the product lifecycle
US FDA-inspected GMP facilities
Other Services
Connect with our scientific experts for your drug discovery, development, and manufacturing needs
We understand that clear communication is essential to successful collaborations, and that's why we have a dedicated team that is always ready to help you. Whether you have questions about our services, want to discuss a potential partnership, or simply want to learn more about our company, we're here to help.
Our team of experts is dedicated to providing personalised solutions tailored to your unique needs. So, please don't hesitate to reach out to us. We look forward to hearing from you and helping you achieve your business goals.
Learning Resources

NOVEMBER 16, 2023
The importance of business continuity planning in CRDMO industry
Both natural and unnatural catastrophic events inflict negative consequences due to the ever-increasing interconnectedness of the global economy. Those consequences are certain to last for longer duration. e.g.; The Covid-19 pandemic is still having a negative impact on the global economy. Maintaining continuity is critical for all businesses, but perhaps no othe...
Read More
HPAPI Molecule from Development to Market
The global Highly Potent Active Pharmaceutical Ingredients (HPAPI) market is expected to reach USD 26.84 Billion by 2023 from USD 17.72 Billion in 2018, at a CAGR 8.7%....
Read More
Physiochemical Characterization Services
Backed by our strong chemistry, we enable “Finger-print” protein structure and functional characterization for proteins from naked proteins to hyperglycosylated or derivatized proteins. ...
Read More
Designing a novel methodology for the development of a macrocyclic peptide molecule for both formulation development and pre-clinical studies
Background: An innovator company based in UK contacted us to support for the development of macro cyclic peptide molecule for pre-clinical studies. This peptide compound has been identified as potential candidate for the “Vaccination booster for elderly” under the therapeutic category of immunology. The synthesis of this compound posed many challenges such as...
Read MoreAugust 28, 2020
Cu-catalyzed coupling-cyclization in PEG 400 under ultrasound: a highly selective and greener approach towards isocoumarins
The combination of CuI–K2CO3-PEG 400 facilitated the couplingcyclization of o-iodobenzoic acid with terminal alkynes under ultrasound, affording a greener and practical approach towards 3-substituted isocoumarins with remarkable regioselectivity. This inexpensive and Pd and ligand free methodology gave rise to various isocoumarins of potential pharmacological i...
Read More-
April 21, 2026
H2SO4-SiO2 catalyzed synthesis, in silico and in vitro evaluation of quinazoline-based fused tetracyclic derivatives as new inhibitors of PDE4B
The 6,6a-dihydro-5H-isoquinolino[2,3-a]quinazoline-5,7,12-trione (DIQT) derivatives (that belong to the quinazoline-based fused tetracyclic class of compounds) though known in the literature, their biological properties have not been explored previously. In the current study, we have explored the phosphodiesterase 4B (PDE4B, an important target for the identifica...
Read More -
January 31, 2025
Development and assessment of a Bcs class II - SGLT2 (Sodium Glucose Cotransporter 2) inhibitor drug in the form of solid lipid Nanoparticles by selecting different lipids, co-surfactants, and manufacturing techniques
Drug Delivery System (DDS) has been used successfully in the past few decades to cure illnesses and enhance health because of its improved systemic circulation and ability to regulate the drug's pharmacological action. As pharmacology and pharmacokinetics advanced, the idea of controlled release emerged, demonstrating the significance of drug release in assessing...
Read More -
January 31, 2025
Development of novel paullone-based PROTACs as anticancer agents
Proteolysis-targeting chimera (PROTACs) represents a promising modality that has gained significant attention for cancer treatment. Using PROTAC technology, we synthesized novel structurally modified paullone-based PROTACs using Cereblon (CRBN) and Von Hippel–Lindau (VHL) E3 ligands....
Read More
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market