From Molecule to Medicine: Integrating Drug Substance and Drug Product for Faster, Smarter Development
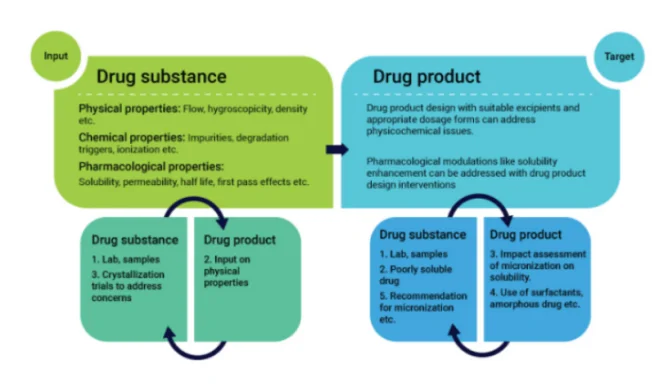
Drug development advances when science, engineering, and operations move as one. The active molecule comes first, but a medicine reaches patients only when that molecule is transformed into a stable, manufacturable, and clinically effective dosage form. This transformation depends on tight integration between drug substance and drug product work streams. When the two tracks run in isolation, projects often slow down at the handover points. When they run together, the feedback loop is faster, development choices are sharper, and the probability of technical success goes up.

The early story starts with the molecule’s identity, purity, polymorph landscape, particle size, and residual solvents. These attributes affect flow, hygroscopicity, and chemical stability. They also shape dissolution, permeability, and exposure. Small changes in crystal form or particle morphology can send a formulation program in very different directions. When teams studying the substance and those engineering the dosage form share data early and often, they match the molecule to the right enabling technology, dosage form, and control strategy with fewer detours.
Integration is not only convenient. It aligns with global expectations for science-based pharmaceutical development. Modern quality guidance encourages defining critical quality attributes for the product, understanding how material attributes and process parameters impact those attributes, and then controlling variability by design rather than by downstream testing. This mindset thrives when analytical science, process chemistry, solid-state characterization, preformulation, and formulation design sit on the same table, work from the same risk register, and iterate on the same development hypothesis.
At a practical level, integrated programs compress time by reducing rework and parallelizing tasks. For example, a salt screen or polymorph study can be guided by biopharmaceutics data and fed directly into early formulation prototypes. Dissolution and permeability results steer particle engineering decisions in real time. Stability signals are traced back to both substance-level and product-level contributors, enabling smarter fixes. Even procurement and logistics benefit when analytical methods, reference standards, and release strategies are harmonized across the API and formulation value chain. For sponsors and patients, the outcome is the same: clearer decision points, faster path to first-in-human, and a more robust platform for later scale-up.
Connect with our scientific experts for your drug discovery, development and manufacturing needs
Overview of Integrated Drug Substance and Drug Product Services
Integrated DS–DP services join process chemistry, analytical development, and solid-state science with preformulation, formulation, and manufacturing sciences. The objective is a single plan that spans from route selection and material control to dosage form design and clinical supply. In this model, a few scientific definitions anchor how decisions are made.
Drug Substance and Drug Product
Drug substance is the active pharmaceutical ingredient in its selected solid form, defined by identity, strength, and purity, along with its solid-state characteristics and residuals. Drug product is the finished dosage form that contains the drug substance together with excipients, designed to deliver the intended dose to the patient with reliable performance.
Critical Quality Attribute, Critical Material Attribute, and Critical Process Parameter
A critical quality attribute is a physical, chemical, biological, or microbiological property of the drug product that must be controlled within limits to ensure quality. A critical material attribute is a property of input materials, including the API, that can influence the critical quality attributes. A critical process parameter is a processing variable that, if not controlled, can cause a change in a critical quality attribute. Integrated teams map these relationships early using risk assessments and experiments, then translate them into a control strategy.
Quality by Design
Quality by design links the target product profile and critical quality attributes to material and process choices. It is easier to practice in an integrated program because the same team can adjust substance form, particle attributes, and formulation architecture in response to the same performance signals.
Biopharmaceutics as the Bridge
The biopharmaceutic profile connects physicochemical attributes to exposure. Solubility, permeability, and dissolution determine how much drug is available at the site of absorption. A shared framework such as the biopharmaceutics classification system helps prioritize enabling strategies and, when appropriate, supports biowaiver paths. Class II drugs typically benefit from solubility or dissolution enhancement. Class III drugs may need permeability-focused strategies and careful consideration of gastrointestinal physiology. Class IV drugs often need combined approaches, with a sharper focus on manufacturability.
Solid-State and Particle Engineering Logic
Polymorph and salt selection are drug substance decisions with drug product consequences. Hygroscopic forms can complicate blending and compression. Needle-like morphologies can hinder flow. Particle size reduction increases surface area and can improve dissolution for low-solubility drugs, yet it may also induce electrostatic handling issues or accelerate degradation. Integration allows trade-offs to be evaluated with both substance and product performance in view.
Dissolution and Release as Sentinels
Dissolution methods are sentinels for oral products. When they are discriminating and robust, they guide scale-up and sometimes enable in vitro–in vivo correlations. In an integrated model, dissolution data inform both particle engineering and formulation design in tandem.
Analytical Life Cycle
Substance and product share analytical anchors such as assay, impurities, and degradation pathways. Methods are designed for life-cycle use so that development, clinical supply, and commercialization are supported without frequent reinvention. Transfer packages are cleaner, investigations close faster, and data integrity improves when a single analytical backbone serves both DS and DP.

Regulatory Line of Sight
A coherent narrative that links substance properties, process understanding, and product performance strengthens the pharmaceutical development section of the dossier. Integration makes the knowledge traceable and shows how understanding increases across the life cycle.
Principles, Definitions, and Scientific Rationale
Quality by Design in Pharmaceutical Development
Quality by design asks developers to link the product’s desired performance to the material attributes and process parameters that can deliver that performance. It encourages early identification of critical quality attributes for the drug product and critical material attributes for the drug substance. The emphasis shifts from testing quality into products to building quality into designs and controls. In an integrated DS–DP model, this becomes practical because both sides of the system are visible and adjustable at the same time.
Biopharmaceutics as the Bridge
The biopharmaceutic profile connects substance properties to clinical performance. Class I substances are high solubility and high permeability. Class II are low solubility and high permeability, often needing solubility or dissolution enhancement. Class III are high solubility and low permeability. Class IV are low solubility and low permeability and usually need multiple enabling tactics. This framework guides selection of particle engineering, solid dispersion, lipid systems, or permeability-focused strategies, and supports eligibility for certain biowaivers when strict criteria are met.
Solid-state and Particle Engineering Logic
Polymorph selection is a drug substance decision with drug product consequences. Hygroscopic forms can disrupt blend uniformity. Needle-like morphologies can clog feeders. Particle size reduction improves dissolution for low-solubility drugs but can create electrostatic handling issues or stability risks. Integration helps evaluate trade-offs early and pick a manufacturable, stable form that supports the required exposure.
Dissolution and Release as Sentinels
Dissolution testing is a sentinel method for oral products. It tracks whether the designed dosage form releases drug at a rate that aligns with intended performance. Apparatus selection, media, and method robustness matter because these data guide scale-up and potential in vitro–in vivo correlations. A joined-up team uses dissolution results to adjust both the particle and the formulation pathway in tandem.

Analytical Life Cycle
Substance and product share analytical anchors. Impurity tracking, residual solvents, and degradation mechanisms inform both API specifications and formulation choices. Validation follows recognized guidelines, and methods are designed to serve development, clinical supply, and commercialization without frequent reinvention. Integrated analytical development reduces inconsistencies at transfer points and speeds investigations when out-of-trend signals appear.
Regulatory Line of Sight
A coherent DS–DP narrative supports the common technical document. Pharmaceutical development sections are stronger when they demonstrate a logical link between material science, process understanding, and product performance. Regulators expect that understanding of variability informs controls and that knowledge grows across the life cycle. Integration makes this knowledge visible and traceable.
What CDMOs typically offer in drug product characterization and testing
Preformulation and Material Understanding
Preformulation begins with a systematic map of solubility across pH, biorelevant media, and cosolvent systems, coupled with pKa determination to locate ionization windows that influence dissolution and absorption. In parallel, solid-state characterization explores polymorphs, salts, and co-crystals, quantifies hygroscopicity and thermal behavior, and establishes crystallinity by techniques such as powder diffraction and differential scanning calorimetry. These data define the landscape of viable forms. Particle engineering then converts form knowledge into performance by tailoring size, shape, and surface through milling, micronization, nanomilling, spray drying, or hot-melt extrusion so that dissolution, flow, and stability move in the right direction. Compatibility studies with excipients under accelerated conditions add a final filter by revealing reactions or physical instabilities that could threaten manufacturability or shelf life.
Formulation Studies Across Dosage Forms
Formulation work translates material understanding into a dosage form that meets the target product profile. For oral solids, the focus is on immediate release or modified release tablets and capsules, including amorphous solid dispersions and lipid-based systems when solubility limits exposure. For parenterals, the emphasis shifts to solutions, suspensions, emulsions, and lyophilized presentations with sterility assurance, particulate control, and container closure integrity. Topicals involve gels, creams, ointments, and transdermal approaches with attention to permeation, rheology, and sensorial attributes. Early clinical enablement relies on fit-for-purpose prototypes that balance speed with performance so that toxicology and first-in-human studies can start while deeper optimization continues.
Analytical Development and Method Validation
Analytical development underpins every decision by creating stability-indicating methods for assay and impurities, supported by forced degradation to prove specificity. Dissolution or release methods are developed with discrimination and robustness so they can guide process changes and support potential correlations to in vivo behavior. Physical tests cover content uniformity, friability, hardness, and disintegration for oral forms; sub-visible particles, osmolality, and extractables for parenterals; and rheology and micro attributes where relevant. Methods are validated with clarity on accuracy, precision, linearity, range, and robustness so that transfers between development, GMP manufacture, and quality control stay smooth.

Stability and Packaging Studies
Stability programs follow ICH conditions for accelerated and long-term storage, with photostability and in-use stability added when the product and clinical context demand it. Data are trended to detect early signals so investigations can close quickly. Container closure studies address compatibility, extractables and leachables risk, and protection against moisture, oxygen, and light. Packaging is selected to support both quality and logistics, with an eye on supply chain constraints and temperature controls for sensitive products.
Manufacturing and Clinical Supply
Manufacturing services translate the laboratory process into GMP batches for clinical studies with a plan for scale-up and, when appropriate, process validation. Clinical packaging and labeling are integrated so that trial supply is coordinated with manufacturing cadence. Documentation and batch records reflect the control strategy developed during quality by design work, making post-approval changes easier to manage later in the life cycle.
Biopharmaceutics and Modeling
Biopharmaceutic assessments connect formulation choices to exposure. Class assignment clarifies which levers matter and whether a biowaiver strategy could be feasible under the rules. Where data allow, mechanistic modeling such as physiologically based pharmacokinetics and in vitro–in vivo correlation helps predict clinical performance, reduce surprises in food effect studies, and prioritize the next experiment. These tools are most informative when they draw from a shared DS–DP dataset with consistent analytical methods.
What Aurigene offers
Aurigene operates as an integrated CRO and CDMO with services that span small molecules and biologics. The company highlights end-to-end development, co-located cross-functional teams, and a legacy of operations that extends over two decades. Integrated DS–DP platforms, one-stop formulation development, stability testing aligned to ICH expectations, and GMP facilities inspected by major agencies create a connected path from substance to product.
- US FDA inspected GMP facilities
- Co-located API and formulation development labs
- Pilot and clinical manufacturing suites for oral, parenteral, and topical dosage forms
- Stability chambers qualified to ICH conditions
- Single-use and intensified platforms for biologics drug substance and drug product operations
- Drug substance route scouting, scale-up, and analytical development
- Solid-state characterization and particle engineering
- Preformulation and biopharmaceutics assessments
- Drug product formulation development for oral, parenteral, and topical products
- Method development and validation, dissolution, and release testing
- ICH stability studies and clinical packaging support
- End-to-end biologics manufacturing covering drug substance and drug product
- Fit-for-purpose strategies for first-in-human enablement
- Integrated DS–DP programs with continuous feedback loops
- BCS-informed development pathways and biowaiver support where applicable
- Amorphous solid dispersions, lipid systems, and micronization for low-solubility drugs
- Lifecycle-ready analytical methods and control strategies
Challenges and Future Outlook
Managing Material Variability
API lots rarely behave identically. Polymorphic drift, residual solvent differences, and subtle particle morphology changes can nudge a process outside its comfort zone. Integrated programs reduce surprises by building acceptance criteria and control ranges that account for both substance and product sensitivities. Continued investment in process analytics and real-time release testing will make this alignment stronger across scales.
Biopharmaceutic Complexity in the Real World
Simulation frameworks and the biopharmaceutics classification system provide structure, yet gastrointestinal physiology varies across populations and with food effects. Dissolution methods that discriminate well in vitro do not always predict exposure under all conditions. The field is moving toward richer biorelevant media, mechanistic absorption modeling, and designs of experiments that couple particle engineering with formulation architecture. As evidence grows, harmonization around in vitro surrogates for clinical performance is likely to expand.

Analytical Life Cycle and Data Integrity
As programs move faster, analytical methods must scale without loss of robustness. Transfer packages need clean traceability, and investigations should resolve quickly. Integrated teams can standardize platforms and keep validation strategies aligned with long-term needs. The direction is toward methods that are stability-indicating, automation-friendly, and fit for continuous verification.
Sustainability and Cost of Goods
Enabling technologies such as hot-melt extrusion, spray drying, and nanomilling can add cost and complexity. Integration helps pick the smallest intervention that achieves the target exposure and manufacturing robustness. As single-use technologies in biologics and intensified processes in small molecules mature, integrated DS–DP choices will increasingly weigh energy, solvent use, and waste as part of the design space.
Global Expectations for Science-Based Development
Regulators encourage a knowledge-driven path. Teams that demonstrate understanding of how material attributes and process parameters impact product quality earn greater flexibility in managing change. Integrated DS–DP operating models make that demonstration more straightforward because the evidence is collected and reasoned within one development story. The expectation is not just compliance but a visible learning system across the life cycle.
Final Perspective
Integration is not a slogan. It is the everyday practice of linking substance properties to product performance and controlling both with clear science. When drug substance and drug product teams share the same development targets, the same risk view, and the same analytical backbone, projects move with fewer surprises. That aligns with good development hygiene and with the direction set by modern pharmaceutical development and biopharmaceutics. In the end, the molecule and the medicine are two sides of one design problem, and they move fastest when solved together.




















