

Project Challenge: Existing formulation (lower strength - 50 mg) was manufactured using direct compression process. This formulation posed poor powder flow and content uniformity issues during scale-up. Development of new strength 150 mg without any process issues was a challenge. Solution design: Preliminary pre-formulation, stress study and PSD impact assessment enabled to understand the molecule nature API Process engineering team helped to achieve appropriate particle size in a short span of time which was used for development

API being needle shaped posed problems when processed by DC process. Hence, new formulation of 150 mg was developed by wet granulation approach. Robust process was developed within 2 months period such that same composition could be applicable for conducting dose ranging study without need for further formula or process optimization The unique attribute of the developed formulation was that it could accommodate 50-200 mg of active ingredient without change in tablet weight. This enabled the customer to quickly fine-tune / finalize the dose based upon clinical study. Activities included, QbD based risk assessment, PSD finalization, Stress studies on API and formulation to understand the drug nature, formula and process design and optimization As the molecule is undergoing clinical trials, the developed formulation provides flexibility with respect to dose and advantage of cost/time which is otherwise needed for development of newer strengths ensuring continuous support for clinical trials
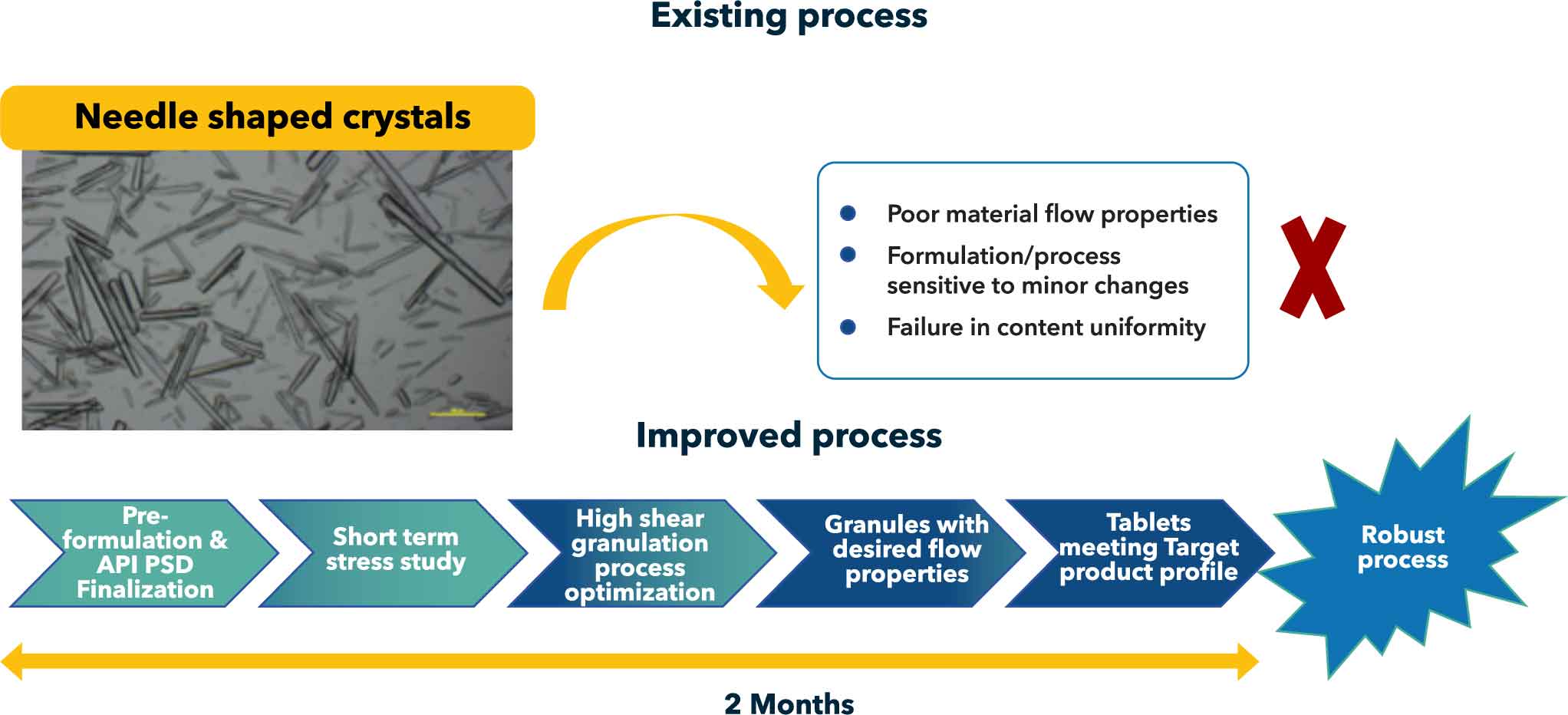
Conclusion: Formulation development and optimization within 2 months with deep scientific understanding and systematic approach. DS-DP integration for on time delivery. Flexible formulation design to accommodate different levels of API with aim of reducing time and cost of development of newly added strengths.
Contact Us
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market
