

Objective: In order to understand the PK properties of a compound and study its effect on animal (dog) model: Studying PK properties of the compound Checking the clearance and bioavailability of the compound Assessing the PK properties to understand if the compound can be used for the clinical trials in humans. Study Design: Species/ Gender: Non-naïve beagle dog/female Study design: Discrete or crossover with 7 days wash out interval; multiple time points and varying concentrations.

The compound was injected through cephalic vein into the plasma; via intravenous or oral route. Concentration of test article (and/or other analytes) in plasma estimated using a fit-for-purpose bioanalytical methods using LC-MS/MS
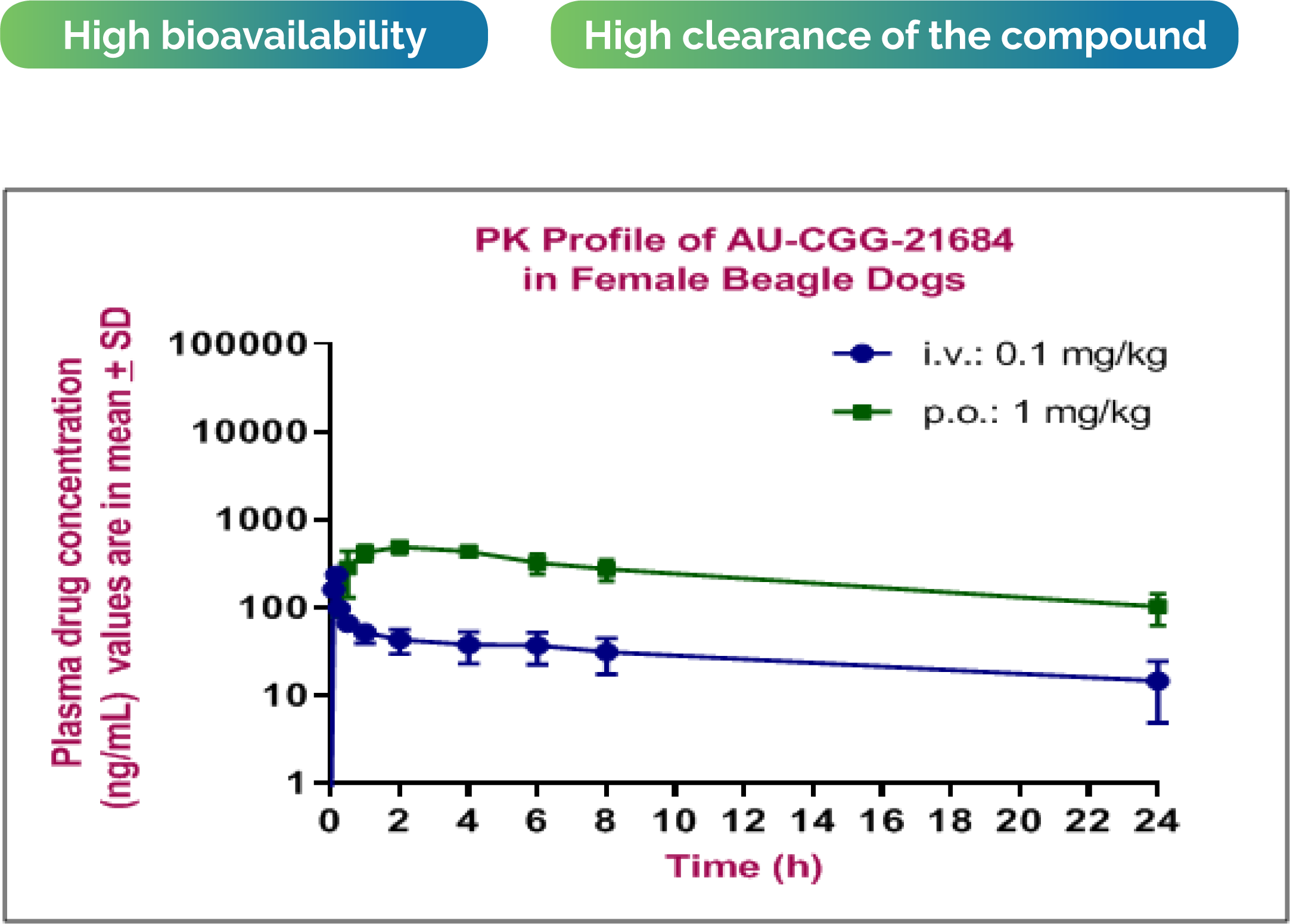
Aurigene solution: PK report with study details: individual & mean time vs. concentration profile was plotted, PK parameters were calculated No test material related observations were noted After intravenous administration at 0.1 mg/kg, the compound showed low volume of distribution and moderate systemic clearance. After oral administration at 1 mg/kg, the compound showed high oral plasma exposure. Observed bioavailability: 87%
Outcomes:
Contact Us
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market
