Aurigene Pharmaceutical Services Limited is a leading contract research, development, and manufacturing organization (CRO/CDMO) and a long-term partner providing end-to-end solutions thereby accelerating innovation. With a strong legacy of services in discovery chemistry, discovery biology, development and manufacturing we are ideally positioned to serve global pharma and specialty companies worldwide by providing truly integrated services.
We handle both small molecule and large-molecule services across various complex technology platforms and niche technologies. We have access to FDA-inspected dedicated R and D centers in India and cGMP sites across India, UK and Mexico. Beyond science, our operations are rooted in the fundamental principles of compliance, information security, quality and sustainability.
Services


Leading CRO/CDMO and a long-term partner
We are uniquely positioned to serve our customers and accelerate their journey to the clinic from discovery to commercialization by nurturing long-term partnerships.
Read more about our infrastructure to see how we are positioned to provide end-to-end services:
Our Drug Innovation
Services Include

Client Speak

“ I’m very satisfied with the chemistry work at Aurigene, collaborated both chemistry (FTE) and biology (FFS). Technical discussion, weekly updates and meetings were good.”
Testimonial By Biotech, US
“Aurigene is very flexible in allocating FTEs and shipping compounds to the preferred destination. Updates on raw material, reports and LNBs are remarkable. Quick turn around time for solving problems using analytical techniques.”
Testimonial By Large Pharma, APAC
“Based on the FFS experience with Aurigene, it increased the bandwidth from FFS to FTE business. The level of communication and transparency in communication was excellent. Project reports and weekly updates are prepared well and on time. ”
Testimonial By Biotech, US
“Based on the pilot peptide program, we extended an FTE collaboration with Aurigene. The scientific team was excellent in communication and problem-solving. The management was proactive and ready to invest in capability and capacity building.”
Testimonial By Large pharma, US
“Happy with the quality of product, productivity and problem-solving capacity. Proactive communication about shipments helped us to plan our studies efficiently. Aurigene is a one-stop-shop for our discovery and development needs.”
Testimonial By Large pharma, EU
“Happy with the scientific team, hence restarted research collaboration. The team is very responsible, trustable, and reliable. All discovery services are under one roof.”
Testimonial By Biotech, US
JUNE 28, 2022
Neoantigen Specific T cells For Cancer Immunotherapy
An effective anti-tumor immune response in human is marked by presence of T cells reactive against neoantigens. Neoantigens are HLA-bound unique peptides arise from tumor-specific somatic mutations. Neoantigens are highly immunogenic because they are not present in normal tissues and hence bypass central thymic tolerance. The success of immune checkpoin...
Read More
Building successful long-term partnerships with CDMOs from early drug discovery through commercialization
Building successful long-term partnerships with CDMOs from early drug discovery through commercialization Maximizing efficiency in drug research, development, and manufacturing is crucial for turning new innovations into therapeutic and financial benefits. Over the past couple of decades, pharmaceutical companies have increasingly turned to contr...
Read More
Cell Line Development
We enable development of stable and high yielding recombinant Mammalian and Microbial lines. ...
Read More
How the re-designed synthesis of a complex carbohydrate can reduce cycle time
Project challenge: Complex carbohydrate chemistry involving a linear sequence of 10 chemical transformations, unstable intermediates and column chromatographic purications. Solution design: Process research and optimization was performed to develop a robust and scalable process which was implemented on commercial scale. Telescoping of reactions reduced the number...
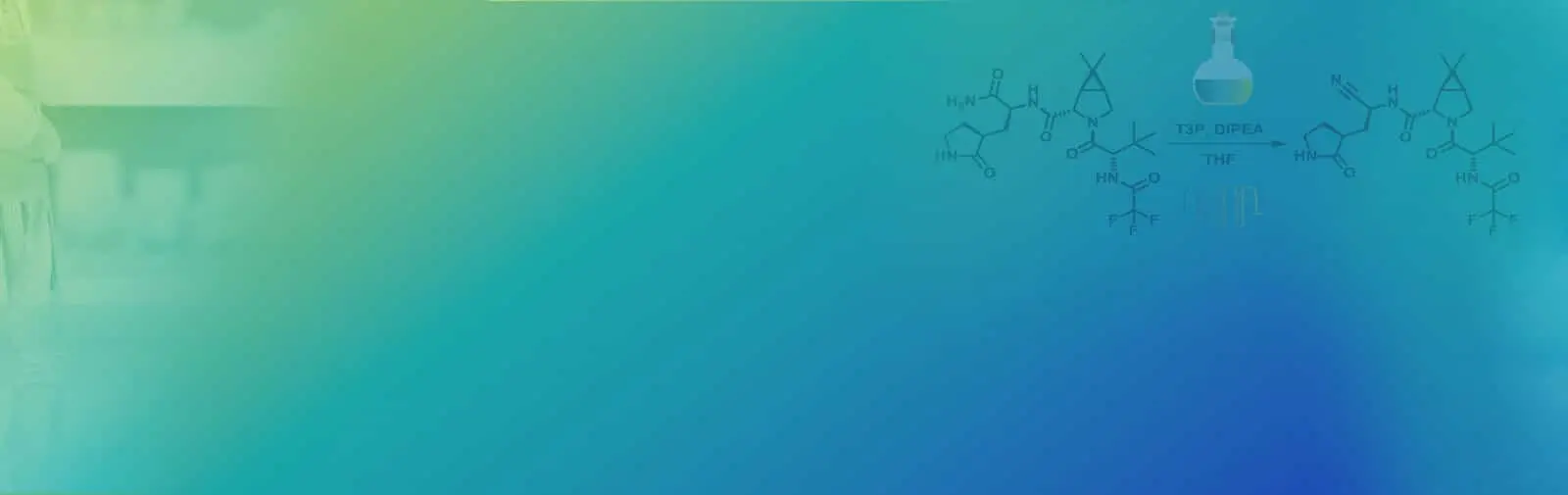
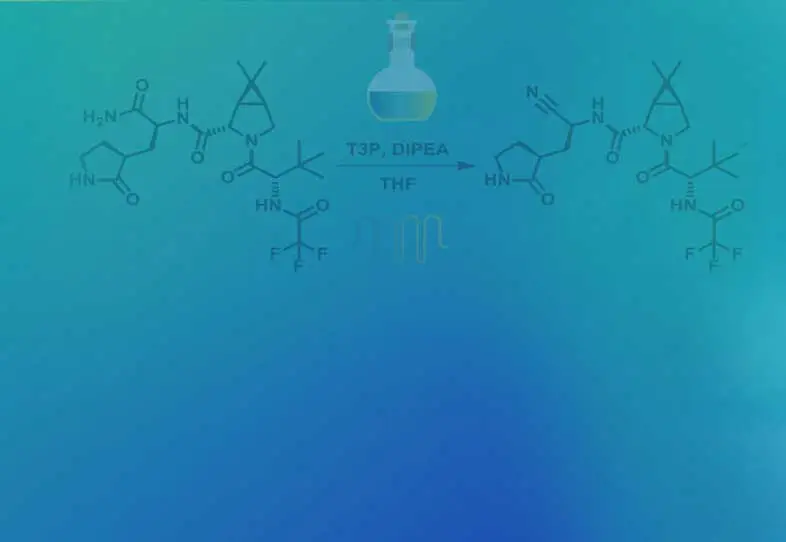
Read MoreAlternate end-game strategies towards Nirmatrelvir synthesis: Defining a continuous flow process for the preparation of an anti-COVID drug
2023
Scalable alternate end-game strategies for the synthesis of the anti-COVID drug molecule Nirmatrelvir (1,PF-07321332) have been described. The first involves a direct synthesis of 1 via amidation of the carboxylic acid 7 (suitably activated as a mixed anhydride with either pivaloyl chloride or T3P) with the ...
Read More-
A Sensitive and a Simple RP-HPLC Method Development and Verification for the Quantitative Estimation of Cholecalciferolin Tablets
2023
Cholecalciferol, also known as Vitamin D3, is widely prescribed for the treatment osteomalacia and osteoporosis [1]. It also plays a key role in calcium and phosphorus homeostasis and skeletal mineralization [2]. IUPAC name of Cholecalciferol is (3β, 5Z, 7E)- 9,10-secocholesta-5,7,10(19)-trien-3-ol, whose molecular weight is 384.64 g/mol and its molecular formul...
Read More -
Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action.
2022
Mutations in MEK1/2 have been described as a resistance mechanism to BRAF/MEK inhibitor treatment. We report the discovery of a novel ATP-competitive MEK1/2 inhibitor with efficacy in wildtype (WT) and mutant MEK12 models. Starting from a HTS hit, we obtained selective, cellularly active ...
Read More -
Synthesis of Anti-covid Drug Nirmatrelvir Using Flow Chemistry
2022
Synthesis of the anti-covid therapeutic Nirmatrelvir by using flow chemistry to enhance efficiency of amide to nitrile conversion in a functionally and Stereochemically Embellished environment. ...
Read More
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market