Integrated
Services


About Aurigene Pharmaceutical Services
Aurigene Pharmaceutical Services Ltd. is an internationally acclaimed contract research, development and manufacturing organization (CDMO) offering integrated solutions to pharmaceutical companies. We combine knowledge and experience with cutting-edge technology to help our clients accelerate their products from lab to market.
Our end-to-end services ranging from discovery chemistry, discovery biology, custom development, manufacturing of clinical compounds and commercial contract manufacturing have been successfully fulfilling the outsourcing needs of pioneering pharma companies for decades. Our world-class R&D centers in India with 650+ scientists are supported by state-of-the-art commercial manufacturing facilities in India, the UK, Mexico and the United States.
Discovery Biology Services
Our range of Discovery Biology Services include
As a leading provider of integrated discovery biology services, our offerings in this domain include biochemistry, cell-based assays, toxicology, microbiology and vitro pharmacology. In collaboration with our clients, our highly-qualified team of skilled professionals has developed meticulously tailored solutions for every phase of discovery research, leading to the discovery of successful clinical candidates.

DMPK (ADME & Bioanalytical)
An expert in the field of ADME - DMPK, we offer a wide range of services in the area of in vitro and/or in vivo ADME - DMPK studies. Our DMPK team is seasoned in integrated drug discovery services and has been associated with 60+ drug discovery programs. To help meet the tight timelines required for drug discovery, we provide accurate and reproducible data with quick turnaround times.
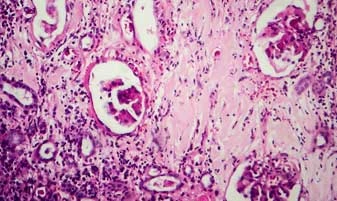
GLP & Non-GMP Toxicology
APSL offers world-class toxicology services with a full range of GLP and Non-GMP toxicology studies supported by clinical, anatomic pathology, formulation analysis etc. Our services include single-dose and repeat dose toxicology studies, genetic toxicology and IND and NDA enabling safety toxicology packages.

In vivo Pharmacology
Our in vivo pharmacology studies are designed to replicate human disease in animals across a range of therapeutic areas that can be customized as per the client’s needs. Consisting of highly experienced scientists with extensive drug discovery backgrounds, this team is well-versed with 1000+ efficacy studies and can handle 600 rodents at a time.

In vitro Biology
Our in vitro biology and screening group offers a wide spectrum of biochemical and cell-based assays to profile small and large molecules. We have in-depth knowledge of different therapeutic areas and target classes enabling us to provide state-of-the-art screening and profiling services.
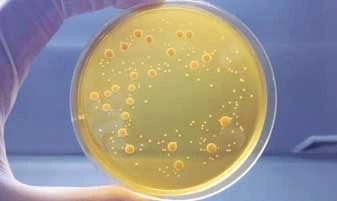
Pharmaceutical Microbiology Testing
With a BSL-2 compliant lab and a separate in-vivo facility, the pharmaceutical microbiology testing department is proficient in executing anti-infective drug discovery projects. It has also participated in several collaborative projects with several pharma companies to discover anti-bacterial and anti-fungal agents.
Aurigene Advantage

Global accreditations Submissions to the US FDA

Plants worldwide International standards Global service provider

Wide range of technologies cGMP compliant facilities Well-equipped facilities Govt. certified labs

20+ years of legacy Experienced team Well-versed with discovery & CMC services Strong and sustainable operations Dedicated manufacturing set up
Connect with our scientific experts for your drug discovery, development, and manufacturing needs
We understand that clear communication is essential to successful collaborations, and that's why we have a dedicated team that is always ready to help you. Whether you have questions about our services, want to discuss a potential partnership, or simply want to learn more about our company, we're here to help.
Our team of experts is dedicated to providing personalised solutions tailored to your unique needs. So, please don't hesitate to reach out to us. We look forward to hearing from you and helping you achieve your business goals.
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market





